Background Information
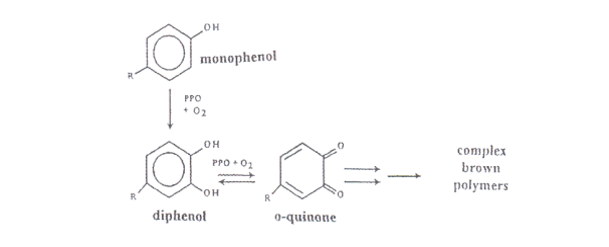
Fruits, vegetables, and even seafood experience enzymatic browning through oxidation. This is due to the rupturing of cell membranes within the fruits. When ruptured, the cells release an enzyme called polyphenoloxidase which when combined with oxygen, creates an abnormally soft texture, and an unappealing brown colour on the surface of the fruit. Polyphenol oxidases are a group of copper proteins that are found in the cytoplasm of ripening plants, where they help with the resistance of microbial and viral infections of plants. The polyphenoloxidase catalyzes two reactions: the o-hydroxylation of monophenols to o-diphenols and the oxidation of o-diphenols to o-quinones¹, which in turn, produce polymers known as melanin. This melanin is responsible for the brown colour that is found in oxidized produce. Oxidized fruits and vegetables are generally unwanted.
There are enzyme inhibitors that can control enzymatic browning. These compounds are classified as reducing agents which remove oxygen, chelating agents which leach out metal compounds, and acidulants which lowers the pH.²
Other ways of slowing the browning of produce are easily achieved through blanching, freezing, and changing the pH level. The activity level of polyphenoloxidases is highly dependent on the pH level of the produce. By lowering the pH to 4.0, the enzyme activity is highly inhibited. The use of different acids all help to lower the pH level of the produce.
There are enzyme inhibitors that can control enzymatic browning. These compounds are classified as reducing agents which remove oxygen, chelating agents which leach out metal compounds, and acidulants which lowers the pH.²
Other ways of slowing the browning of produce are easily achieved through blanching, freezing, and changing the pH level. The activity level of polyphenoloxidases is highly dependent on the pH level of the produce. By lowering the pH to 4.0, the enzyme activity is highly inhibited. The use of different acids all help to lower the pH level of the produce.
Purpose
Everybody in our world eats fruits and vegetables; it's one of the four essential food groups, and it is essential in maintaining good health and survival. Almost every fruit or vegetable goes through the process of oxidation. This allows your fruits to soften and turn into an unappealing brown shade, making them unappetizing to yourself and to your guests or clients. Preventing browning can save a lot of money and fresh produce; up to 40% of the produce grown in the UK is thrown out due to blemishes that are often caused by enzymatic browning.² The food industry also wastes a lot of 'ugly' produce because they strive to serve beautiful dishes. Knowing how properly to slow down the oxidation of fruits can make the food that is eaten more physically appealing and will create a better environment by reducing the amount of fresh produce that is wasted.
Question
Which of the 5 solutions (pure lemon juice, pure orange juice, white vinegar, 1 tbs of salt dissolved in 1 cup of water, and sprite) will slow the time that it takes for apples, pears, and avocados to turn into the darkest shade of brown?
Independent Variable
The independent variable in this experiment is the five different kinds of liquids in which the cubes of fruit are submerged in.
Dependent Variable
The dependent variable in this experiment is the varying shades of brown that the fruit cubes become when exposed to the air.
Controlled Variables
The controlled variables include the room temperature (19 degrees Celsius), the kind of fruit that is being used (red delicious apples, Chinese pears, Haas avocados), the size of the fruit cubes (2 cm), the amount of time that the fruits are exposed to the liquids, the amount of time that the fruit cubes are exposed to the air, and the colour chart that is used to measure the shade of brown that the cube has become.