Qualitative Data
Throughout the whole experiment, the shade of brown in which the apples, pears, and avocados developed into was recorded and compared with each other. We noticed that with Sprite, orange juice, vinegar, and our control, the development of the brown colour was gradual and increased by similar amounts each time. However, the salt water solution and the lemon juice stopped the development of the brown colour for the first two hours and then suddenly stopped to work, turning the fruits into a much darker shade by the sixth hour. This process was mimicked in both the second trial and the third trial. When touching the fruits, they all slightly softened as time went on, but the lemon juice soaked fruits seemed to be a little bit crisper.
Quantitative Data
|
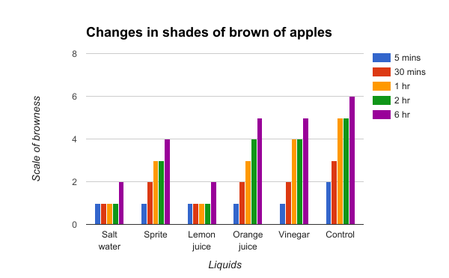
Figure 1 shows that for the apples, the salt water and lemon juice slowed down the oxidation of the fruits compared to the other solutions. All the other liquids except for the lemon juice and salt water showed gradual increase in the shade of brown as time progressed. Even after multiple trials, the lemon juice and salt water remained having the same results; both liquids were able to delay the enzymatic browning until the 2 hr point.
|
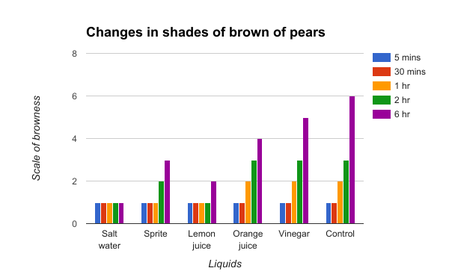
The results in figure 1 and figure 2 all very similar due to the fact that both these fruits have similar levels of polyphenol oxidase. The salt water solution showed drastic results in slowing the oxidation as the shade of brown remained the same at the 5 minute point and also the 6 hour point. The lemon juice and the sprite were able to hold off the oxidation process before beginning to show changes at the 2 hour and 6 hour mark respectively. The orange juice and the vinegar had the exact same results as the control, but at the 6 hour mark, it outperformed the other 2 liquids.
|
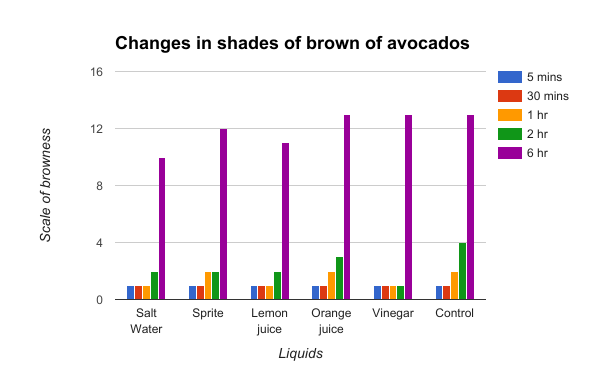 Figure 3
Figure 3
Figure 3 shows the relatively low shades of brown throughout the first 2 hrs among the 6 different solutions. However, the brown pigments increase dramatically at the 6 hour point due to the large amounts of polyphenol oxidase present in avocados as compared to the apple or pear cubes. Although the shades of brown the avocados experienced at the 6 hour mark were very close,our results verified our hypothesis as the salt water solution did indeed slow it down the most. As shown in the graph, the orange juice and vinegar did help to slow down the enzymatic browning, but at the end it remained as the same shade of brown as the control which proves its ineffectiveness in retarding discoloration.
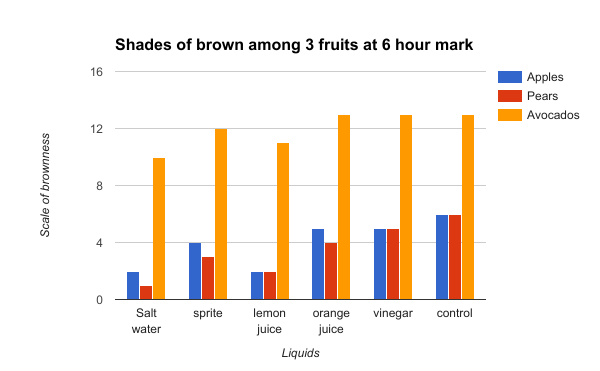 Figure 4
Figure 4
Figure 4 shows the comparison of the three fruits at the 6 hour mark. From the graph, we can see that the one liquid that consistently delayed the enzymatic browning was the salt and water solution. Lemon juice comes in a close second as it slows down the oxidation of fruits at the same rates as the saltwater. Sprite comes in as the third best at delaying the browning based on the average of the fruits. Orange juice and vinegar did not show much change compared to the control, as it often stayed as the same shades.
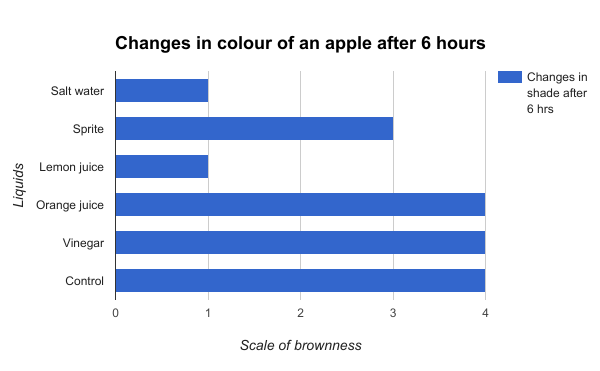 Figure 5
Figure 5
Figure 5 shows the changes in colour that occurred in the apples after 6 hours. The changes were calculated by the equation (final shade-initial shade). It is evident from the graph that the salt water and lemon juice solution only caused an increase by one shade after 6 hours, whereas the orange juice, vinegar, and the control increased by 4 shades after 6 hours.
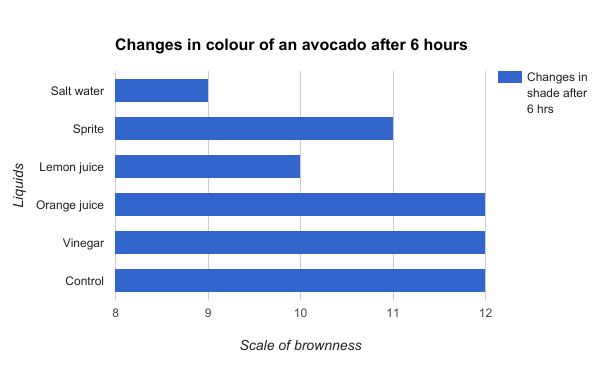 Figure 6
Figure 6
Figure 6 shows the changes in color of an avocado after 6 hours. This time, the salt water solution resulted an increase of 8 shades of the fruit, while the lemon juice resulted in an increase of 10 shades. These two liquids did significantly better than the orange juice,vinegar, and control which resulted in an increase of 12 shades.
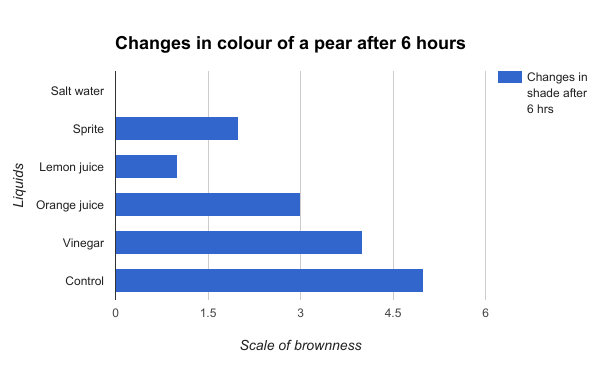 Figure 7
Figure 7
Figure 7 shows the changes in colour of a pear after 6 hours. The salt water has no bar because it showed no increase in shades after 6 hours. The orange juice and vinegar had a better effect on pears in slowing down the oxidation compared to the control.
Reasons For the Performance of the Liquids
The saltwater solution performed the best out of the five different liquids, despite it having a pH level of 8. This puts it into the neutral category, which is unlike the four other liquids that were tested. The saltwater was the most effective in slowing the browning in fruits because the chloride ions in the salt inhibit the polyphenol oxidase (PPO) enzymes from working properly and the water cuts off the enzyme's access to oxygen. The second most effective liquid that was tested was lemon juice. Lemon juice has a pH level of 2 which makes it extremely acidic. The PPO enzymes are inhibited by acids as well, so the more acidic the liquid, the more effective it is in slowing the oxidation process. The lemon juice did not perform as well as the saltwater because unlike the saltwater, it did not cloak the fruit in water, cutting off the oxygen supply of the fruit. The third most effective liquid was the Sprite because it contains a large amount of citric acid. This, in turn, lowers the pH level of Sprite to 3, making it an acid. As well, the large amounts of sugar in the soda completely coat the cubes of fruit, stopping the exposure to air. The fourth most effective liquid was the orange juice. The pH level of orange juice is 3, making it an acid. It is not as strong as lemon juice and does not have an ingredient to coat the fruit, which explains why is it not as effective as the liquids mentioned earlier. The liquid that performed the worst out of all of them was the vinegar. Despite being as low as lemon juice on the pH scale, vinegar did very poorly due to its properties as an acid. The acetic acid in vinegar is a not an antioxidant, unlike the acids listed above, so it does not cut off the oxygen that is trying to react with the fruit. The pH level of the liquids plays a large role in the slowing of the oxidation process, but there are always some exceptions, such as saltwater and vinegar.